Bringing innovative ADC and SMDC medicines to cancer patients around the world
Founded in 2018, CanWell Pharma is led by several highly accomplished veterans who have an average of 25 years of experience in drug discovery and development in both U.S. and China biopharmaceutical industries. CanWell is dedicated to developing its proprietary first-in-class and best-in-class oncology products to address difficult-to-treat advanced cancers. CanWell is developing a rich R&D pipelines under our technology platforms.
Our Mission
Our mission at CanWell is to develop innovative treatments that harness the power of the immune system to fight cancer. We are devoted to leveraging the latest advances to further explore and develop a variety of innovative therapies. With a focus on collaboration, innovation and patient-centered care, we strive to be at the forefront of cancer therapy and bring new hope to patients and their families.
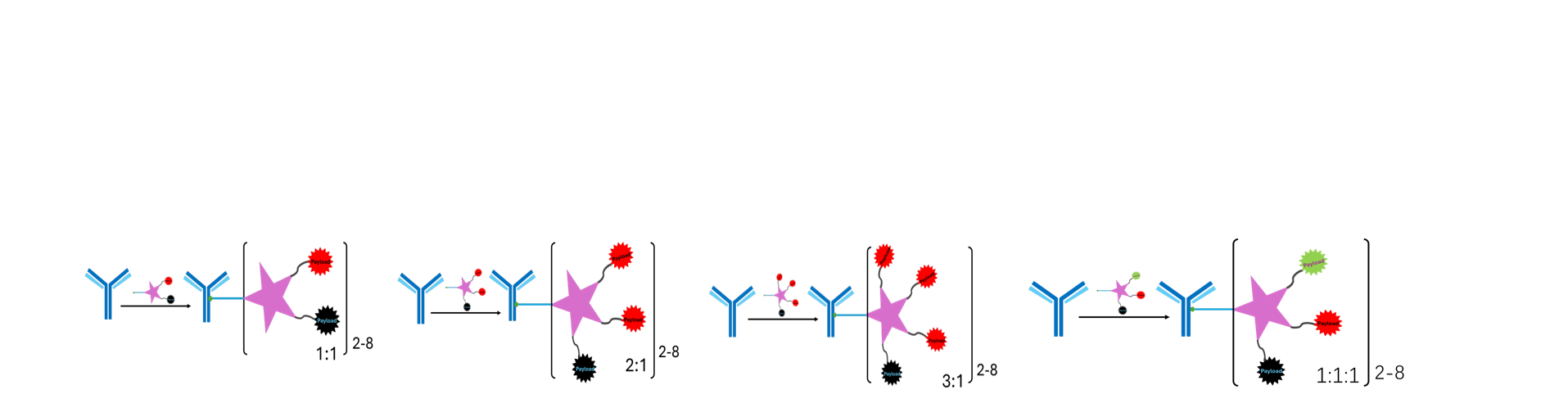
Antibody Drug Conjugate
Small Molecule Drug Conjugate
Latest News
CanWell Pharma will present three R&D posters at the AACR 2026 Annual Meeting.
On April 2, 2026, CanWell Pharma announced that the research progress of three of its self-developed pipelines has been selected for the poster presentation session at the 2026 American Association for Cancer Research (AACR) Annual Meeting. The AACR Annual Meeting is one of the most influential acad…
CanWell Pharma Announces FDA Clearance of the IND for CAN016, a Dual-Payload ADC Targeting HER2, for ADC Pre-Treated Solid Tumors
IND approval from the FDA allows CanWell Pharma to assess the safety, tolerability and pharmacokinetics of CAN016 in a Phase I clinical study CAN016, a novel antibody-drug conjugate (ADC), is expected to overcome the resistance mechanisms that limit the efficacy of conventional, single-payloa…
CanWell Pharma Announces Participation in the AACR Annual Meeting 2026.
San Diego, CA – CanWell Pharma is pleased to announce its participation in the AACR Annual Meeting 2026, taking place from April 17–22, 2026, at the San Diego Convention Center. The AACR Annual Meeting is the focal point of the cancer research community, where scientists, clinicians, other health ca…
CANWELL Pharma Received U.S. FDA Orphan Drug Designation for CAN1012 in the Treatment of Soft Tissue Sarcoma
March 3, 2026, the U.S. Food and Drug Administration (FDA) granted Orphan Drug Designation (ODD) to CAN1012, the company’s proprietary IFNα-biased TLR7 agonist, for the treatment of Soft Tissue Sarcoma (STS). CAN1012 is a novel small-molecule IFNα-biased Toll-like receptor 7 (TLR7) agonist developed…